ISLAMABAD: In a historic development for public health and patient safety, the Drug Regulatory Authority of Pakistan (DRAP) has granted formal registration for Tadalafil, a widely used medication for sexual health disorders, to four Pakistani pharmaceutical companies. This marks a significant turning point in the availability of quality-assured, legal treatments for erectile dysfunction and related conditions in Pakistan.
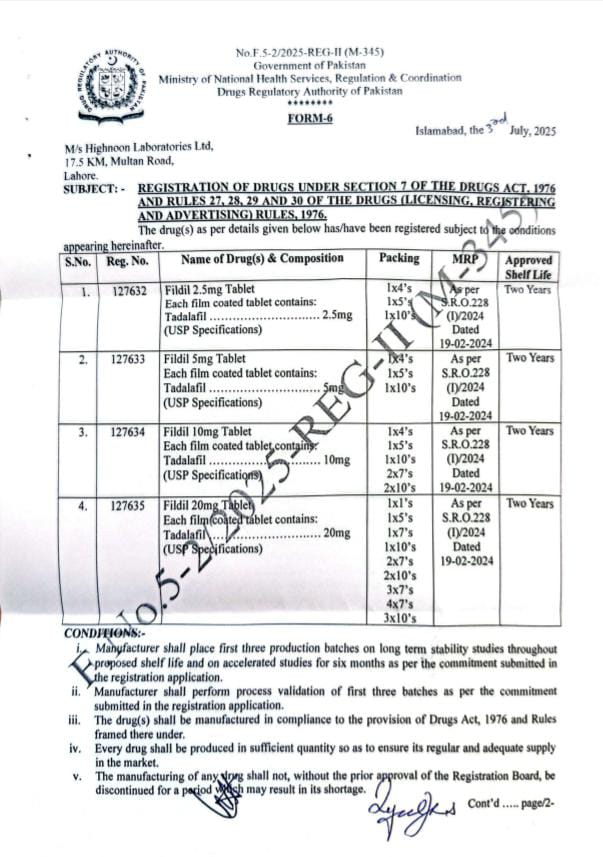
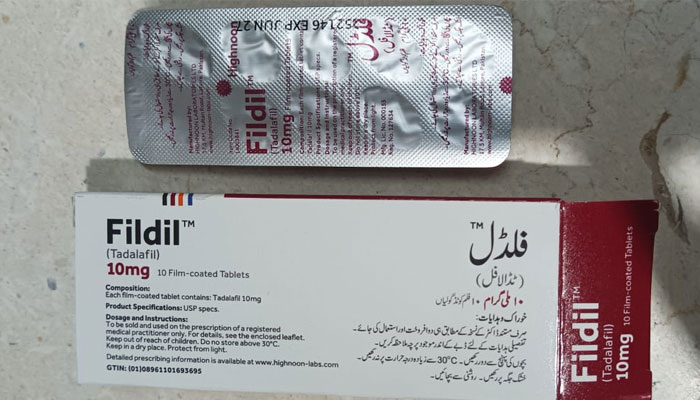
Among the companies granted registration, Highnoon Pharmaceuticals, Lahore, has become the first to officially launch its Tadalafil product, branded as “Fildil”, in the local market. This launch ensures safe, regulated access to a treatment that was previously available only through unregistered, smuggled, or counterfeit sources.
“This is a highly welcome and historic move that protects public health and curbs the circulation of illegal, substandard drugs,” said Noor Mahar, Advocate and President of the Pakistan Drug Lawyer Forum (PDLF). “It’s a major success for DRAP and a blessing for patients — especially those suffering from diabetes, heart disease, and erectile dysfunction.”
Background: From Smuggled Pills to Legal Medication
For years, Pakistan’s sexual health medicine market was flooded with over 30 counterfeit and unregistered brands, largely smuggled from India or manufactured locally without regulatory oversight. The situation was especially concerning in provinces like Khyber Pakhtunkhwa, Sindh, and Balochistan, where these products were sold openly, often in unsafe formulations such as illegal syrups and unregulated pills.
Until now, Sildenafil Citrate (Viagra and its generics) was the only registered medication available for erectile dysfunction. The introduction of Tadalafil — known for its longer duration of action and better tolerance in patients with chronic illnesses — now offers a clinically effective and safe alternative.
Medical Community Reaction
The move has been praised by healthcare professionals across Pakistan. Urologists and general physicians have highlighted that the registration of Tadalafil provides a reliable, safer choice, particularly for patients who cannot tolerate Sildenafil or have coexisting conditions such as diabetes or cardiovascular diseases.
Pharmaceutical Sector Applauds DRAP
The pharmaceutical industry has also welcomed DRAP’s decision. The move is being seen as a milestone in strengthening Pakistan’s drug regulatory regime and combating the illegal medicine trade.
The CEO of DRAP and the leadership team have received accolades from the pharmaceutical and legal communities for taking concrete steps toward patient safety and quality assurance.